Molar mass of nh34/13/2023 The molar mass of ammonia (NH3) is 14.007 + 3(1.008) = 17. Calculate the molecular mass of- NH3, CO2, HCl, Cl2 - Get the answer to this question and access a vast question bank that is tailored for students. 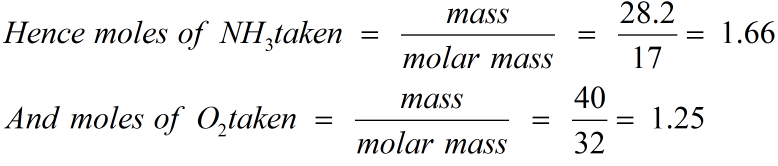
Step 4: Calculate the grams of ammonia using the molar mass. The balanced chemical reaction equation says that for every 3 moles of H 2 there are 2 moles of NH 3 produced. Calculate the molecular mass of- NH3 CO2 HCl Cl2 - Get the answer to this question and access a vast question bank that is tailored for students. Step 3: Use the molar ratios to find the moles on ammonia produced Compound X has a molar mass of 579.36 g-mol and the following composition: element mass phosphorus 26.73 nitrogen chlorine 12.09 0 61.18 Write the molecular formula of X. 
so for H2 it is 1.008(2) = 2.016 gram/mole: In a particular reaction, 0.575 g of NH3 forms. Click hereto get an answer to your question Calculate the molecular mass of NH3. In a particular reaction, 0.575 g of NH3 forms. How many molecules (not moles) of NH3 are produced from 5.01×104 g of H2 How many oxygen atoms are in 2. The molar mass of hydrogen is 1.008 g/mole. we need the molar mass of NH3 N 14 grams H 1 gram 14 + 3 17 grams per mole of NH3 mass produced 7.0217 119.34 g How many grams of H2 are needed to produce 12.51 g of NH3. Step 2: Turn the 25 g of hydrogen gas (H2) into moles by using the molar mass. Click here to get an answer to your question Calculate the molar mass of Nh3. Therefore, the molecular mass of NH3 14 + 3x1 14 + 3 17 By definition, mole molecular mass in grams. Now we have six hydrogens on the right so we need six on the left meaning we would need to write a "3" coefficient in front of the H 2 like this: (The number 3 should be in subscript) The atomic masses of N 14, H 1. Molar mass of Ammonia (NH 3) 14.0067 g/mol + (3× 1.00794) g/mol 17.03052 g/mol 17.03052 g/mol. Normality is equal to the molarity of the solution when molar mass is equal to the equivalent mass of the solute.Step 1: Write a balanced chemical reaction equationįor N 2 + H 2 → NH 3 we have two nitrogens on the left so we need two on the right, meaning we need to add a "2" coefficient in front of NH 3 like this: N 2 + H 2 → 2NH 3. Frameworks for Ultra-High Uptake and Selective Separation of Ammonia. In other words, molar mass is the sum of the mass of all the atoms found in one moles worth of a substance. succeeded in the molecular electrochemical reductive splitting of. It can only be equal to or greater than the molarity of the solution. Molar mass can be defined as mass per mole. Note that normality of a solution is never less than molarity of the solution. Molar mass of NH3 (g) is 17.03052 0.00041 g/mol Get control of 2022 Track your food intake, exercise, sleep and meditation for free. 
The expression used to calculate the percentage composition in w/v is 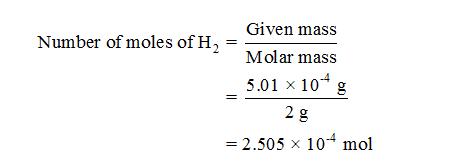
Hint: Weight/volume (w/v) % of a component means the amount of the component dissolved in 100 mL of the solution.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
